May 2023 Global Innovative Drug Report
This May 2023 Global Innovative Drug Report offers a comprehensive overview of the latest advancements in the pharmaceutical industry and an analysis of each selected drug. Within the report, we delve into various aspects of each drug, including its snapshot, R&D status, active patents, mechanisms of action, clinical trials, competitive landscape, and other crucial factors.
Download it now to learn about:
- The first approved drugs in May
- New drug analysis
- Global drugs under expedited review pathway in May
- Analysis of selected ERP drugs

May 2023 Global Innovative Drug Report Overview
1.) Drugs Approved in May
In May 2023, a total of 18 drugs received global approval, representing a diverse range of therapeutic categories. These include:
- 4 Small molecule drugs (SMD)
- 4 Monoclonal antibodies (mAb)
- 2 Prophylactic vaccines
- 2 Allergen extracts
- 1 Blood component
- 1 Gene therapy
- 1 Bispecific antibody
- 1 Enzyme
- 1 Fusion protein
- 1 Synthetic peptide
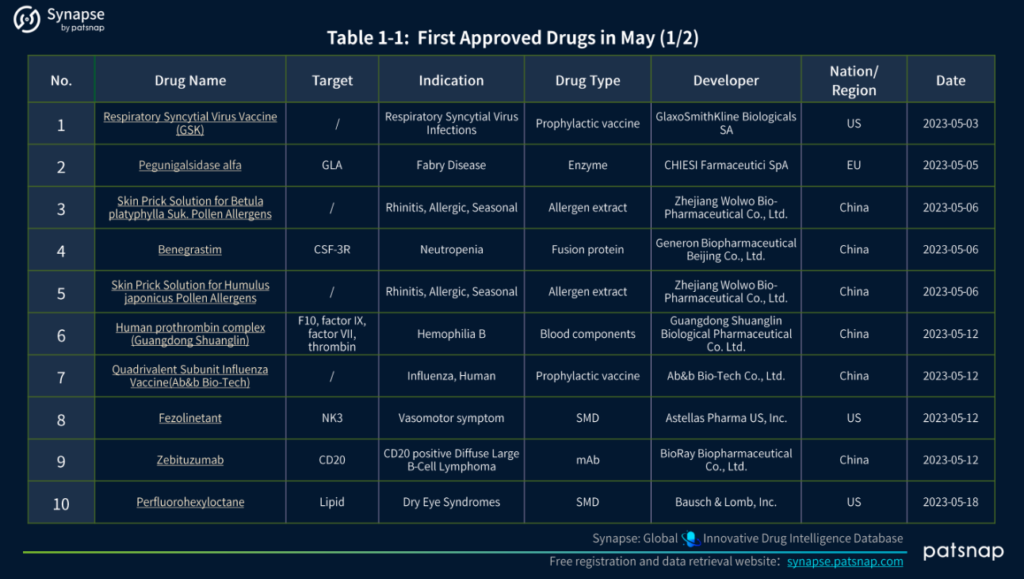
2.) Comprehensive Analysis of Newly Introduced Medications
This report also offers a comprehensive analysis of five recently approved drugs, providing valuable insights into their profiles and impact. Let’s delve into the in-depth analysis of Benegrastim, one of the featured drugs, which encompasses the following comprehensive details:



3.) Global Drugs Under Expedited Review Pathway in May
In May 2023, there were 57 drugs worldwide undergoing Expedited Review pathways. These included:
- 32 Orphan Drug designations
- 12 Fast Track designations
- 5 Priority reviews
- 4 Breakthrough Therapy designations
- 2 Regenerative Medicine Advanced Therapy designations
- 1 Innovative Licensing and Access Pathway (ILAP)
- 1 Accelerated Approval
These designations and pathways aim to accelerate the evaluation and approval process, ensuring timely access to innovative treatments for patients in need.

4.) Analysis of Selected ERP Drugs
Within this report, we selected five drugs under Expedited Review pathways for detailed analysis. Now, let’s explore the report content focused on Spesolimab, which provides an in-depth examination of the following key details:




Gain Access to the Comprehensive Report for FREE – Download Now! If you aren’t registered for Synapse (registration is required to download the report), click here to register for free.
PatSnap Synapse Database: An Overview and Key Features
Discover the innovative PatSnap Synapse database – an AI-powered platform that provides intuitive interfaces, curated content, and access to massive information sources. With integrated access to company data, diseases, targets, clinical studies, biological and chemical entities, our platform offers a powerful search and association experience. Sign up for free today and experience the benefits for yourself!
R&D Decision Makers
Our mission is to empower R&D decision makers with swift access to accurate and connected data, facilitating their understanding of emerging technology trends, competitive landscapes, and partnership opportunities. By providing comprehensive insights, our platform helps decision makers to navigate and steer the direction of innovation with confidence.
Business Development Professionals
We offer a comprehensive database that covers the drug pipeline and investment history for over 320,000 life science organizations. This invaluable resource allows you to make informed decisions regarding potential partnerships or acquisition targets with confidence. Gain access to our extensive database and unlock the insights you need to drive your business forward.
Pharmaceutical Analyst
Our platform is designed to accelerate the research process for pharmaceutical analysts by leveraging a wealth of connected data, including drug approvals, clinical trials, patents, non-patent literature, and news. With Synapse, users gain a comprehensive 360-degree view of the competitive and technological landscape, empowering them to make informed decisions quickly and efficiently. Discover the power of Synapse and revolutionize your research today.
Copyright Statement: This report is the sole property of Patsnap and is protected under copyright laws. Any reproduction, excerpting, or other use of this report without explicit authorization from Patsnap is strictly prohibited. Authorized products must be used within the scope of authorization and must include a clear indication of the source. Patsnap reserves the right to investigate any violations of this statement and pursue legal action as necessary. For inquiries regarding authorization, please contact [email protected].
