Synapse Visitor–AI Pharmaceutical Intelligence Platform
On April 20, PatSnap, the world’s leading SaaS provider for innovation intelligence, announced the launch of its latest version of the intelligence platform, Synapse Visitor. This version includes advanced search functions for drugs and pharmaceutical organizations, now publicly accessible for the first time. Users can explore these features without prior registration, making searching easier and more accessible than ever before.
Synapse is an innovative Life Sciences Platform that uses AI technology to connect Drug Discovery information with Drug Development insights. This allows professionals in the pharmaceutical industry to make better-informed decisions throughout the entire innovation cycle.
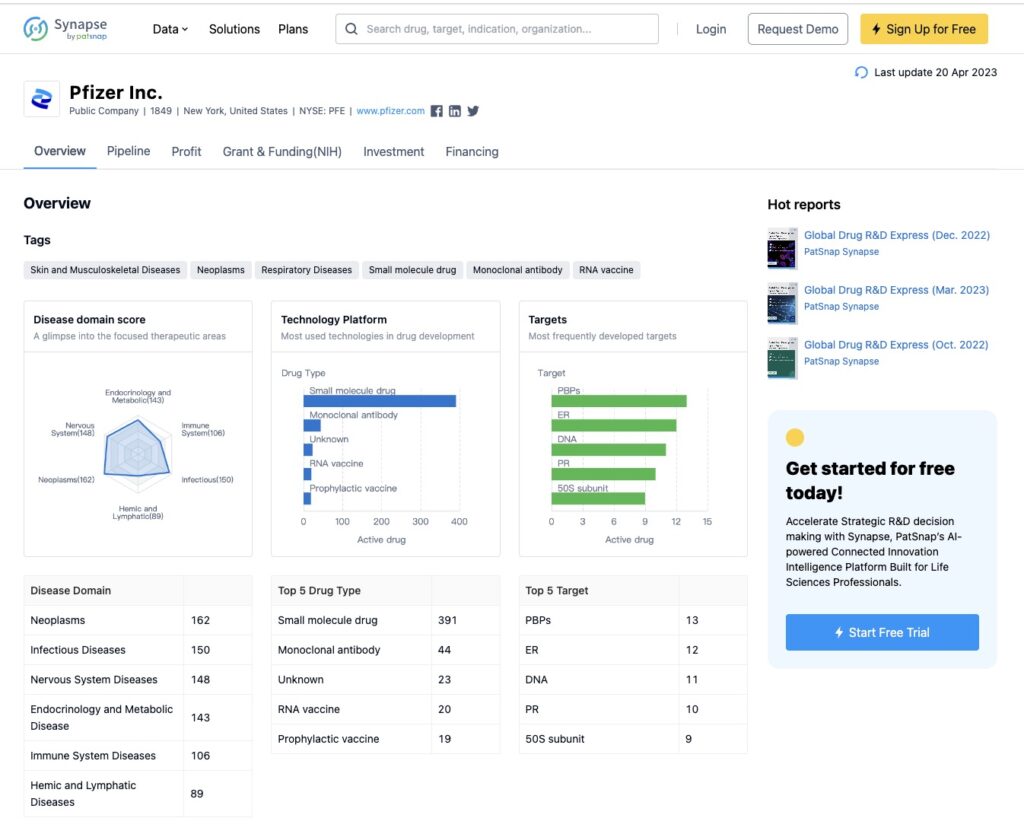
With a vast database of millions of free data points from over 150 jurisdictions, Synapse provides access to over 72,000 new drugs, 800,000 clinical trials, 6 million pharmaceutical patents, 62 million literature sources, 360,000 pharmaceutical organizations, and more. This wealth of information is available to users to aid them in their research and decision-making processes.
One of the key benefits of Synapse is its extensive database of drugs, particularly those in their preclinical or early developmental stages, which helps to mitigate potential risks for professionals in the pharmaceutical industry. “We want to ensure that we are only developing drugs that are efficiently managed, reducing the risks of failure at the later stages of development,” said one of our key clients, the head of R&D. By having access to this valuable information, professionals can make more informed decisions and focus their efforts on drugs with a higher potential for success, ultimately saving time and resources.
Available on Synapse’s website as a free solution, key features of Synapse Visitor include:
1.) Drug Advanced Search:
Quickly and accurately identify drugs based on specific criteria of interest. The search function covers global drug candidates and marketed drugs, and mines preclinical R&D intelligence in patents and literature to identify early competitive risks.
2.) Clinical Progress:
Screen clinical trials by drugs, institutions, targets (multi-target), indications, clinical trial phase, and other conditions. Synapse collates data from 18 registries, including CT.gov and WHO, giving users a global view of the clinical landscape.
3.) Playbook:
Access multiple data sets with a single search, including Due Diligence and Find Organizations. This feature provides comprehensive and visualized insights into the organization’s R&D preferences, drug pipeline, investments, financing, and more.
4.) Literature Search:
With over 62 million literature records in the life sciences field, users can easily screen literature by drugs, targets, indications, authors, and other criteria. This feature allows for efficient and targeted research, saving time and increasing productivity.
To learn more about PatSnap and its AI-powered pharmaceutical intelligence platform, visit synapse.patsnap.com. For a step-by-step tutorial on how to leverage Synapse and access pharmaceutical data, please visit https://www.youtube.com/@SynapsebyPatSnap.
About PatSnap: Founded in 2007, PatSnap is the company behind the world’s leading AI-powered innovation intelligence platform. PatSnap provides global businesses with a connected, easy-to-use platform that helps them make better decisions in the innovation process. Customers are innovators across multiple industry sectors, including agriculture and chemicals, consumer goods, food and beverage, life sciences, automotive, oil and gas, professional services, aviation and aerospace, and education.
Media Contact:
Antasha Durbin
Email: [email protected]
Your recommended content
-

Patsnap Surpasses US$100 Million in Annual Recurring Revenue
Category: Article | Category: News/PR
Wednesday, June 12, 2024
Patsnap has reached a significant milestone of achieving $100M in Annual Recurring Revenue (ARR), marking an impressive 20% year-over-year growth in 2023. This milestone highlights the massive and meaningful value our platform brings to over 12,000 IP and R&D teams across 50 countries, driving efficiency, productivity, and collaboration.
-

Transforming Patent Intelligence: Patsnap Partners with BizInt Smart Charts
Category: News/PR | Category: Partnership
Thursday, February 1, 2024
In a significant development for patent professionals worldwide, Patsnap has collaborated with BizInt Smart Charts to empower users with streamlined data integration. This strategic partnership facilitates the direct export of Patsnap data into BizInt Smart Charts for Patents, simplifying the transition from patent search to comprehensive analysis.
-
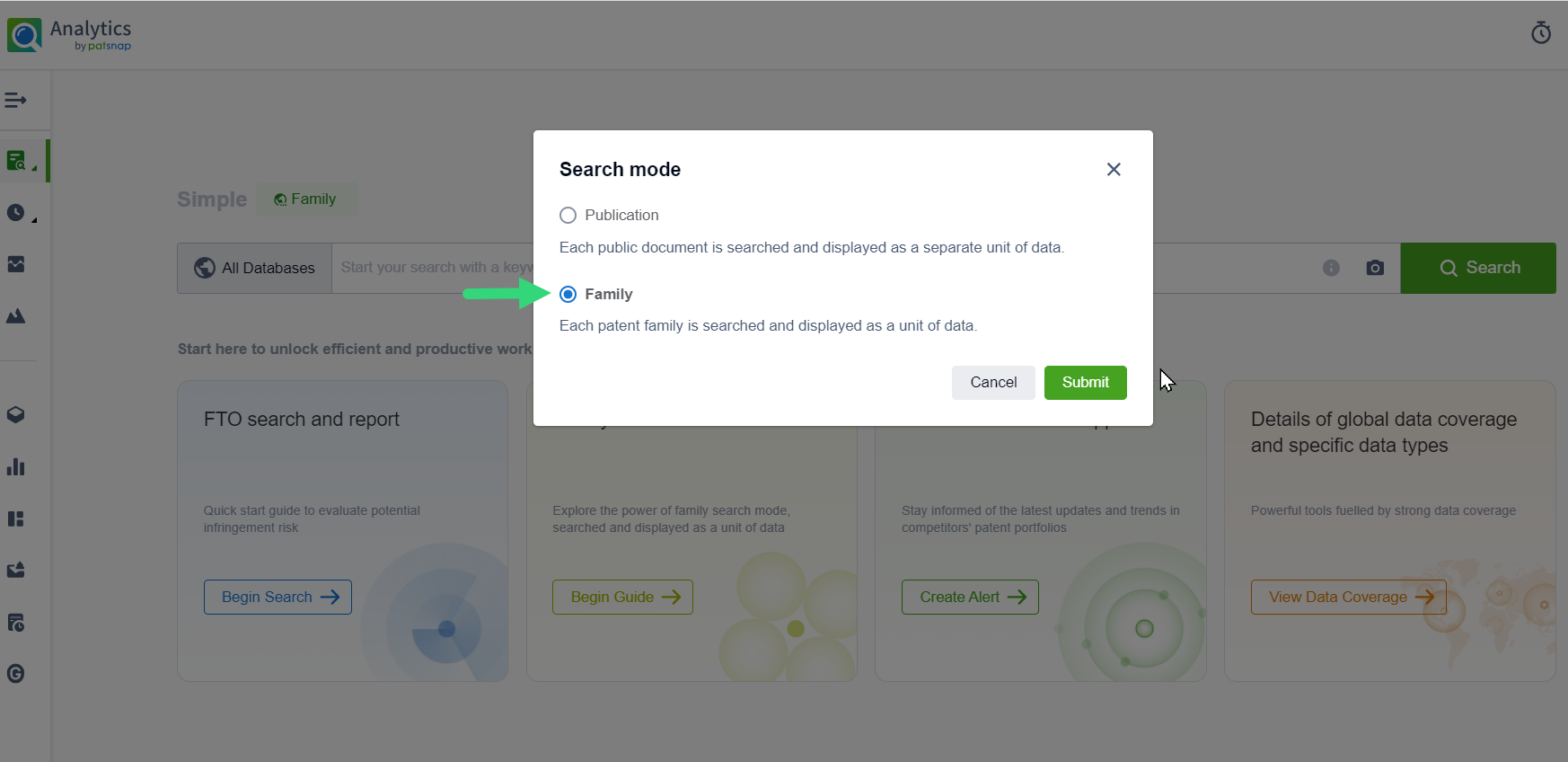
Patsnap’s Analytics Launches Family Mode Searching
Friday, December 8, 2023
Introducing Family Search Mode! Explore patent innovation like never before with Patsnap's Analytics, treating each patent family as a data unit for enhanced insights into interconnected technologies