Intellectual property in life sciences
The risk (and opportunity) presented by patent cliffs indicates a need for increased data coverage in patent intelligence platforms that provide insight into the life sciences.
A 54% decrease in bio and pharma IP activity
Intellectual property has been a cornerstone in the long-term strategy goals for many organizations, and the past few years have been no different. As the impact of COVID-19 continues to take the global economy by storm, many organizations are forced to reconsider their strategies with respect to acquisitions, research and development, and intellectual property. In 2019, biotechnology and pharmaceutical companies spent $306B USD on IP-related activities. In contrast, the same organizations spent only $140B USD in 2020—indicating a 54% decrease in IP-related activity (source: Lexology). Although this slump may alarm a few, with last year’s losses creating over $1.4T USD in excess funds, 2021 is set to be a memorable year for intellectual property trends in the Life Sciences industry.
Despite many positive indicators, however, patent cliffs (the term for when patent rights to drugs expire, opening the way for generics and other competition) continue to be a significant challenge in a highly regulated industry like life sciences. and while there are options like patent extensions or supplementary protection certificates, the risk (and opportunity) presented by patent cliffs indicates a need for increased data coverage in patent intelligence platforms that provide insight into the life sciences. This article examines what patentability looks like in the life sciences, the specific challenges and opportunities presented by patent cliffs, and how patent searching is being shaped by recent trends.
Intellectual property in the life sciences industry is central to the development and commercialization of these innovations. For an idea to be patentable, it must be novel, nonobvious, and useful; all of which are evaluated on a case-by-case basis by territorial patent offices. In the life sciences, this could mean a unique use for a particular nucleotide, protein sequence, or a new laboratory method to an age-old scientific technique (Molecular Biotechnology). Patents encourage industrial and economic growth by releasing intellectual property to the public, thereby allowing companies to invent using patents, through licensing, and around patents, by learning from the information disclosed in the patents and then the creating novel ideas and concepts.
Areas that will shape investment in the life sciences industry in 2021 are oncology, cell and gene therapies and rare diseases. Monoclonal antibodies (mAb) and recombinant DNA technologies are pivotal to these sectors and provide the most potential for preventive and curative solutions. These technologies require enormous investment, so the availability of patent protection, including the effects of recent case law, for new developments is important to understand for firms considering making these investments.
As an example, Monoclonal antibodies are laboratory-made proteins that mimic the immune system’s ability to fight off harmful pathogens. They are crucial to certain cancer or autoimmune drugs (such as rituximab), and can be used in treatment of SARS-CoV-2 (bamlanivimab) (Harvard College v. Canada, concerned the patentability of higher life forms, specifically the ‘Harvard oncomouse’, a transgenic mouse that promoted a cancer gene. The main concern in this litigation was the patenting of the “founder” mouse, which was considered a higher life form and is not a patentable subject in Canada. Ultimately, this case was ruled in favor of the patent after an amendment which omitted the concept that higher life forms cannot be ‘manufactured’ or a ‘composition of matter’. This case teaches that transgenic animals are exclusively a vessel for new innovations and cannot be treated as one entity.
Another case relevant to the patentability of genetic material was that of Molecular Pathology v. Myriad Genetics, whereby linkage analysis correlated BRCA1 and BRCA2 to an increased risk of breast and ovarian cancer. To further test this hypothesis, Myriad’s scientists isolated the genes to determine specific chromosomal mutations that infer risk. This isolation was deemed unpatentable as entire genes cannot be protected exclusively based on isolation from the human body. Again, this case shows the difficulties of navigating potential opportunities because of the specificities surrounding definitions of patentability and regulation differences between countries who enforce patent law.
The Economic Challenge of Patent Cliffs
In addition to technological challenges in the Life Sciences industry, there also remains the economical challenge of patent cliffs. A patent cliff refers to the phenomenon wherein a number of patents representing a large portion of sales revenue are set to expire at the same time, thereby allowing competition to enter and drive down the price (Springerplus). The patent cliff is particularly noticeable when blockbuster products, those which exceed $1B USD in sales annually, expire at the same time allowing competitors to enter the market and remove the company’s market share for that indication. With the innovator company guaranteed to lose a significant market share ten to twelve years after every market entry, the patent cliff could mean over $100B USD in sales erosion. As a result of long research and development timelines and large startup costs in a highly regulated industry, the patent cliff is particularly a pain point for larger biotechnology and pharmaceutical companies. Conversely, the patent cliff presents opportunity for small and medium sized (SMB) companies and generic manufacturers to replace blockbuster products due to the shift from a monopolized market to a competition-based one, where price is a defining feature. For smaller companies, the ‘first-to-file’ race could be vital to the overall company health, making efficient patent searching a priority. Effectiveness and efficiency are crucial for companies in earlier stages of research and development and strategic filing of patents as well as intelligent patent cliff prediction could mean survival or demise of smaller organizations.
To mitigate the looming challenge of patent cliffs, Supplementary Protection Certificates (SPC) are provided as an intellectual property right that serves as an extension to a patent expiration for up to five years. While SPCs are common in European countries, Patent Term Extensions (PTE) take precedence in North America. Both SPC and PTE offer a method to extend patents to compensate the regulatory approval delay involved in patent filing and market authorization. The patent on which these special circumstances can be applied for are primarily biologically active agents or human medicaments, particularly those which need to undergo many stages of clinical trials. These extension certificates allow for prolonged exclusivity and act as a reward for innovators who have endured the long regulatory hurdles. Additionally, SPC manufacturing waiver regulation offers EU-based companies to manufacture generic or biosimilars, if in markets where there is not any IP protection for the drug. This allows EU-based manufacturers to gain global competitiveness and to encourage the generics and biosimilars market.
Patent Search and Innovation Intelligence Platforms in the Life Sciences
In an everchanging global economy, intellectual property remains high priority to many organizations. The life sciences industry is highly competitive as emerging technologies, personalized medicine and targeted therapies promise hopeful cures to detrimental illnesses, new outlooks on agricultural implications and industrial improvements. Now more than ever, ease of sequence searching, and patent synthesis could mean significant financial as well as strategical gains. In an industry where there is growth in the biosimilar and generic market, data and market exclusivity are becoming increasingly vital. A SaaS patent company like PatSnap offers a helping hand in staying ahead of the ‘patent race’ by providing extensive data coverage for biological sequences in major jurisdictions which offer SPC or PTE extensions. By identifying patent timelines and potential regulatory hurdles, organizations will be able to reduce costs and refocus on innovation.
Author

Piya Jain
Project Manager, Life Sciences, PatSnap
Your recommended content
-

Patsnap Surpasses US$100 Million in Annual Recurring Revenue
Category: Article | Category: News/PR
Wednesday, June 12, 2024
Patsnap has reached a significant milestone of achieving $100M in Annual Recurring Revenue (ARR), marking an impressive 20% year-over-year growth in 2023. This milestone highlights the massive and meaningful value our platform brings to over 12,000 IP and R&D teams across 50 countries, driving efficiency, productivity, and collaboration.
-

Introducing Hiro, an AI assistant built for IP and R&D workflows
Category: AI advancements | Category: AI development | Category: AI-tools | Category: Article | Category: artificial intelligence
Tuesday, May 14, 2024
Powered by Patsnap’s industry-specific LLM, Hiro is designed to streamline IP and R&D workflows from ideation to product launch. With its robust AI capabilities, Hiro brings a new level of efficiency, precision, and security to tasks that were once time-consuming and labor-intensive.What sets Hiro apart is that it draws from our large language model that’s been trained on market-leading patent records, academic papers, and proprietary innovation data. This ensures we deliver more accurate and reliable results for every prompt.
-
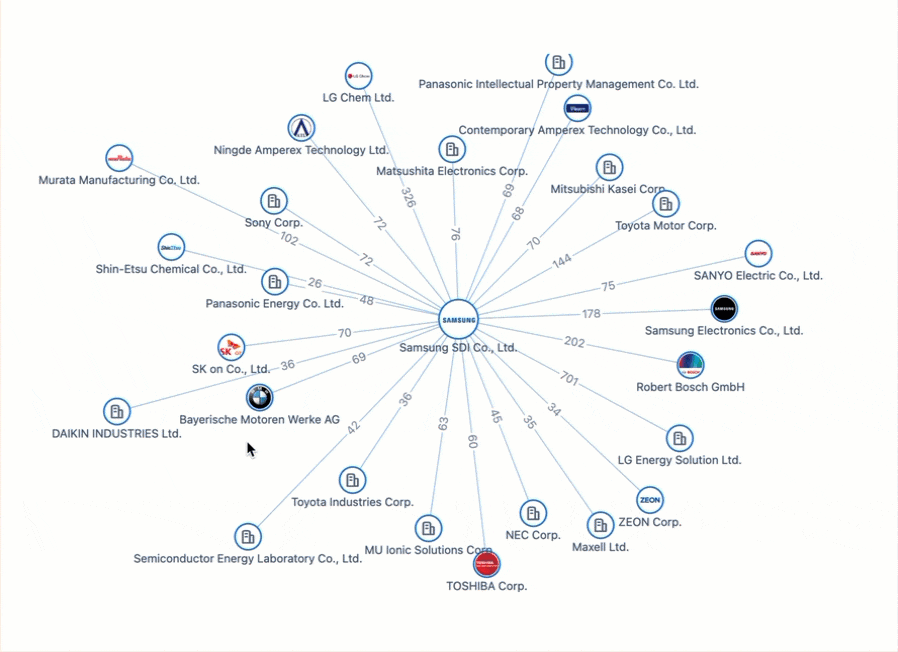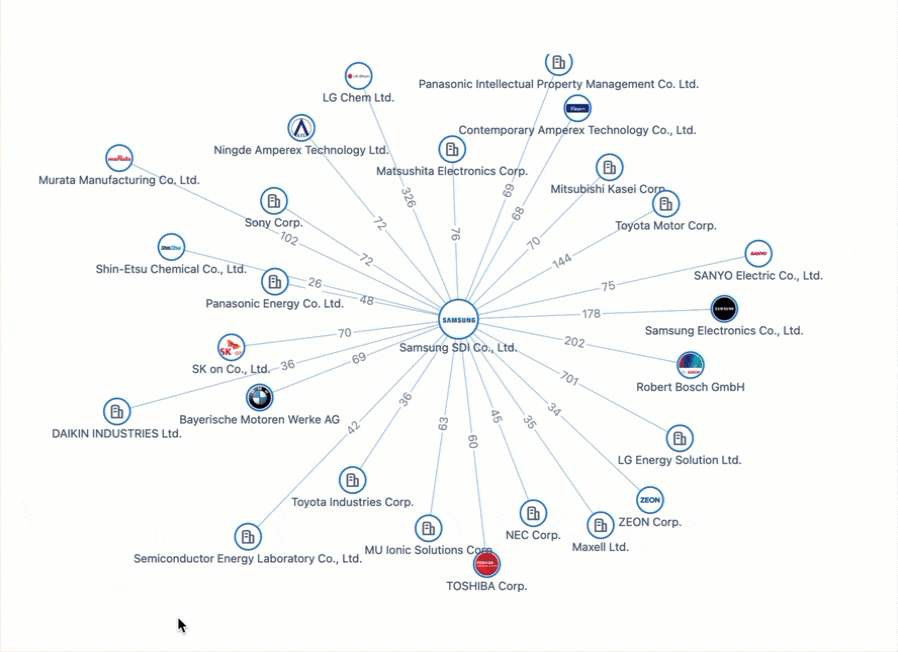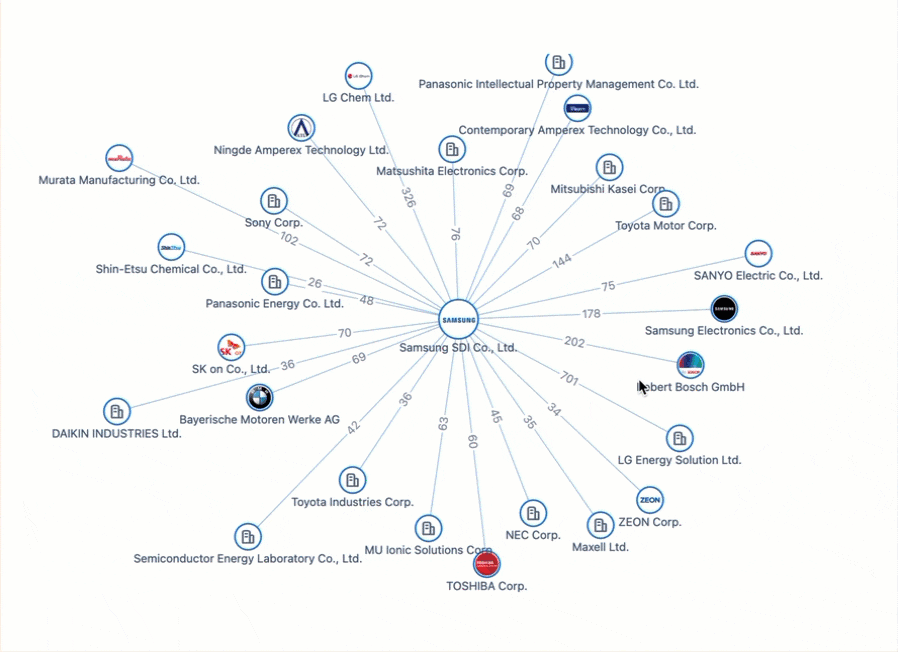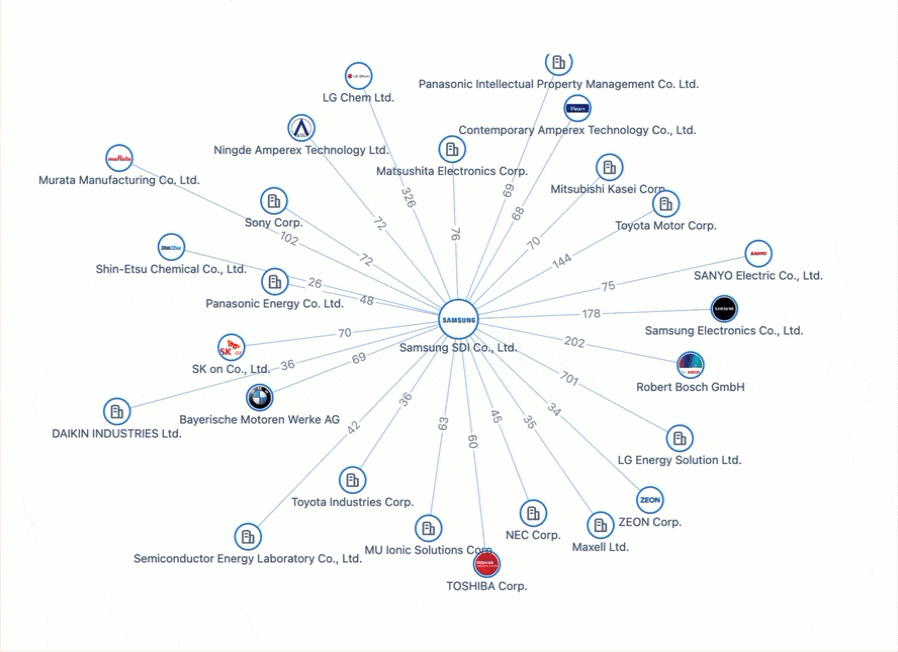
Powering the Future of Electric Vehicles: The Battle for Battery Innovation and Patents
Category: Article | Category: battery technology | Category: electric vehicle | Category: EV | Category: lithium ion | Category: lithium ion battery | Category: NEV | Category: new energy vehicles
Monday, April 22, 2024
In the ever-evolving landscape of innovation, the electric vehicle (EV) industry stands as a beacon of technological transformation. As we explore the patents propelling the EV revolution, Apple's venture serves as a poignant example of the challenges even industry giants face in this competitive arena. Join us on a journey through the global patent landscape, where the quest for superior power solutions unfolds, and where the true pioneers of the EV revolution are making their mark.